DataBytes Newsletter
Interdisciplinary Team Develops Cadaveric Perfusion Model for Lifesaving Biomedical Device Testing
Injuries to the carotid artery of the neck are particularly common during combat, as it is in one of the few areas not protected by armor. Penetrating neck injuries also occur due to stab wounds and automobile accidents. The carotid is a vital artery that supplies blood to the brain, which requires a continuous supply of blood. In the case that the carotid gets lacerated and begins to hemorrhage, the individual has minutes to live unless medical care is administered. The current protocol for this type of injury is to have somebody pinch the vessel to stop the bleeding, which requires constant attention from the medic, and prevents them from treating other injuries or other individuals that might need emergent care. Currently there are no occlusive devices on the market that target this critical region of the neck. This is a significant problem due to the high mortality rate of wounds to the carotid arteries and jugular veins that runs through this region.
A team of Cedarville University biomedical engineering students (Austin Ballentine ’19, Caleb Williams ’19, Devan Kienitz ’19, and Bennett Stouffer ’19) took on the development of a neck occlusion device after Lauren Edmonson ’19, an allied health major, came to Cedarville Professor Dr. Timothy Norman, and suggested the project. Lauren realized the deficit in the available treatment options for this injury while undergoing training as a volunteer EMT.
A year after the completion of the project and continued development, one design, the mechanical Arterial Restrictive Clamp (ARC), emerged as a clear winner and a viable option as a neck occlusion device for military and civilian use. The ARC team, consisting of Dr. Norman, and Edmonson , Ballentine and Williams, was joined by surgeon Dr. Jeff Williams of Durango Colorado. Dr. Williams is a fully trained general surgeon and practicing plastic, hand, and craniofacial surgeon. He is also active in training special forces operators and military medics in trauma care.
As with any medical device looking to reach the marketplace, considerable testing is required. For a device designed to stop arterial blood flow, clinical studies are not possible. This is where the cadaveric perfusion model comes in. Following the work of researchers at the University of Southern California who developed the model, Dr. Norman assembled a team of investigators that could create a cadaveric perfusion model at Cedarville University. In addition to the ARC team, the team recruited help anatomy professor Dr. Melissa Burns and medtronic perfusion specialists Joseph Agoston from Cincinnati Ohio. The team also received generous help from Dr. Angelia Mickle and Karen Callan of Cedarville University’s School of Nursing and Scott Huck from marketing and communications. Donations were also received from Kettering Heath Network. “Putting this team together was certainly God led, as the people, equipment, and help that we needed seemed to fall into place,” remarked Dr. Norman.
The cadaveric profusion model (Fig. 1) is designed to provide simulated blood flow in a cadaver to allow for the testing of medical devices and surgical repair techniques that otherwise would not be possible due to the nature of the procedures. In this model, synthetic blood is pumped from a reservoir into the femoral artery via a Medtronic BioMedicus 550 centrifugal pump. Synthetic blood flow rate and arterial and venous pressures are monitored to ensure near physiological pressure and flow conditions. Once the proper conditions are established in the cadaver, the carotid artery was 100% lacerated. In successive tests, the surrounding soft tissue was lacerated in increasing length and at each length, the ARC was tested for its effectiveness in tamponading blood flow. Testing outcomes were remarkable and proved the effectiveness for the ARC as a lifesaving device. “The application of the ARC has the potential to make an almost unsurvivable injury treatable for military and civilian patients even in mass casualty situations,” remarked Edmondson, now a 2nd lieutenant in the United States Air Force, who is an M.D. candidate at the Uniformed Services University of Health Sciences. The device is currently in the patent process.
“This work was only possible through the generous help of many and from blessings from the Lord. We were especially thankful to Wright State Human Donor Program for supplying a donor that suited the needs of the project well. The loss of this loved one was instrumental in the testing of the lifesaving device,” remarked Dr. Norman.
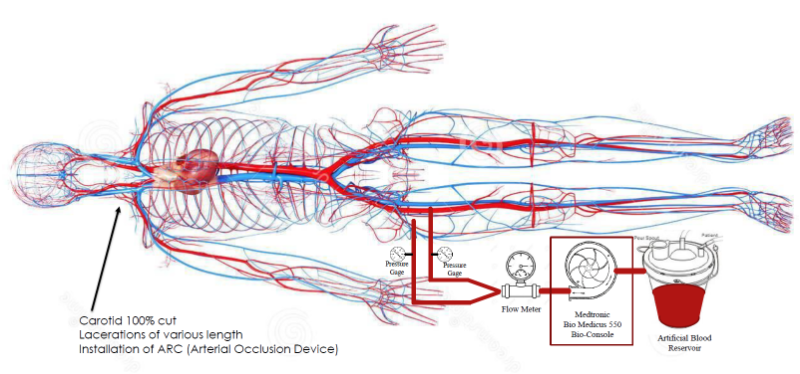
Fig. 1. Cadaveric Perfusion Model for testing the arterial occlusion device.
(Circulatory System Dreamstime.com 26592808)


















